|
ΑΝΑΚΟΙΝΩΣΗ (PRESENTATION)
Βιβλιογραφική αναφορά (Citation)
|
|
IDEA Medical Newsletter
2009:3,5. Available at https://www.idea-lab.gr/newslet20090305.htm
|
 Interasma Regional Meeting Interasma Regional Meeting
Marrakech,
27-30 November 2003
Evaluation of
a new in vitro test (IAST®) for the screening of allergy in
asthmatic patients
A. Papaconstantinou, A. Makrygianni,
U. Spyrakou
Medical Diagnostic Laboratories
(Athens)
Purpose
of the study: The clinical evaluation of a new in vitro
screening test for the diagnosis of respiratory allergy in asthmatic
patients.
Patients
and methods: 290 unselected adult and paediatric patients
with asthma were studied (118 males and 172 females) aged 1 to 84 years
(mean: 35,1 years). The diagnosis of
asthma was based on history, clinical examination and (in persons able to
perform) lung function tests.
Skin prick tests
were performed in 286 patients with the standard set of Anallergo (Florence,
Italy). Any reaction with a wheal
being ≥ 5 mm in diameter after 20 minutes was counted as positive.
Specific IgE
determinations for 15 common inhalants were done with the enzymatic
allergo-sorbent test (EAS) of Bio-Line (Brussels, Belgium).
Finally all
patients were examined with the new in vitro test IAST® of Eurolab
(Athens, Greece). IAST®
(Inhalation Allergy Screen Test) is an immunoenzymatic method which uses as
solid phase a balanced mixture of pollens with code name POL and another
mixture of inhalant allergens with code PER.
The manufacturer does not supply the exact composition of the
allergens. The method is quantitative
and the normal reference value is 0-35 AU/L.
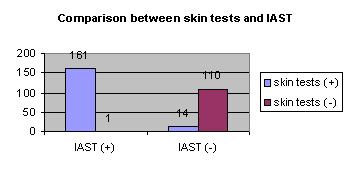 Results: At least one positive
skin test had 175 patients and 161 of them had an IAST® value >
35 AU/L. Of the 111 patients who were
skin tests negative only one had an IAST® value > 35 AU/L. 14 patients had normal IAST value with one
or more positive or weakly positive skin tests. Results: At least one positive
skin test had 175 patients and 161 of them had an IAST® value >
35 AU/L. Of the 111 patients who were
skin tests negative only one had an IAST® value > 35 AU/L. 14 patients had normal IAST value with one
or more positive or weakly positive skin tests.
164 patients had at
least one positive specific IgE and only one of them had an IAST®
value ≤ 35 AU/L. 126 samples
were negative with both IAST® and Bio-Line methods.
The geometric means
of IAST® positive and negative samples were 876,3 and 12,6 AU/L
respectively. Higher IAST®
values observed in patients with high specific IgE to individual allergens
rather than in multiple sensitivity.
In positive blood
samples relative disagreement between IAST® and Bio-Line EAS was
found in 6 cases (e.g. positive in both indicators with positive specific IgE
only for pollen).
The statistical
analysis of results of the present study shows that IAST® has
(depending on the reference method) sensitivity 92-99,4%, specificity
99-100%, positive predictive value 99,4- 100% and negative
predictive value 88,7-99,2%. 100% and negative
predictive value 88,7-99,2%.
Conclusions:
IAST® was shown to be highly sensitive
and specific in differentiating asthmatics who are sensitised to common
inhalants from those who are not. It
is a simple, reliable method in first intention for the physician who faces a
putative respiratory allergy. It is
quantitative and distinguishes pollen allergy from this due to other
inhalants. Moreover it is an excellent
biological confirmation for a patient with questionable negative skin tests.
►Επιστροφή στην πρώτη σελίδα του IDEA Medical Newsletter
Προειδοποίηση
Το περιεχόμενο του IDEA MEDICAL NEWSLETTER είναι
επιστημονικό και απευθύνεται σε γιατρούς και λοιπούς επαγγελματίες υγείας. Σε
καμιά περίπτωση δεν πρέπει να χρησιμοποιηθεί από πρόσωπα που δεν έχουν τα
προσόντα νόμιμης άσκησης του ιατρικού επαγγέλματος για οποιαδήποτε
θεραπευτική ή διαγνωστική εφαρμογή.
|